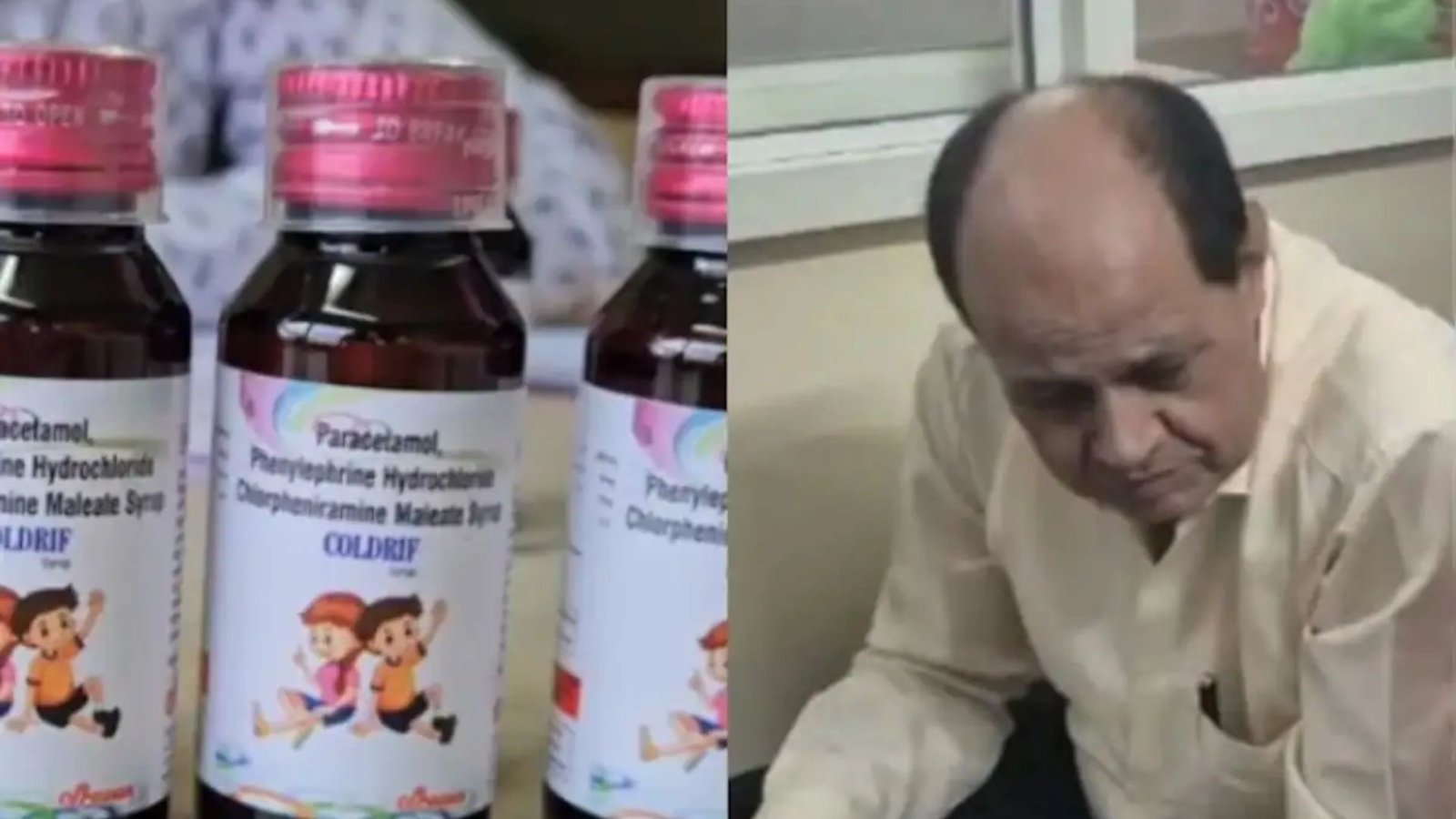
G Ranganathan, the owner of the pharmaceutical company that manufactured the ‘poisonous’ Coldrif cough syrup, has been arrested in connection with the ongoing investigation into the tragic incident. The arrest comes amid mounting scrutiny and public outcry following reports of severe adverse reactions linked to the consumption of the syrup. Authorities have been working diligently to determine the exact cause of the contamination and to hold those responsible accountable.
The Coldrif cough syrup incident has raised serious concerns about the quality control measures within the Indian pharmaceutical industry. Preliminary investigations suggest potential lapses in the manufacturing process, leading to the presence of harmful substances in the syrup. Health officials have issued urgent warnings, advising the public to refrain from using Coldrif cough syrup and to seek immediate medical attention if they experience any adverse symptoms after consuming it.
Investigation Details
A special investigation team (SIT) has been formed to thoroughly probe all aspects of the case, from the sourcing of raw materials to the final distribution of the product. The SIT is comprised of experienced investigators and pharmaceutical experts who are working to gather evidence and identify any regulatory violations. The team is also collaborating with international agencies to gain insights and best practices in ensuring pharmaceutical safety.
The arrest of G Ranganathan marks a significant development in the investigation, signaling the authorities’ commitment to bringing those responsible to justice. Ranganathan is expected to face charges related to negligence, product liability, and potentially criminal offenses depending on the findings of the investigation. The authorities are confident that further arrests may follow as the investigation progresses and more evidence comes to light.
The incident has prompted a nationwide review of pharmaceutical manufacturing practices, with the government announcing stricter regulations and enhanced monitoring mechanisms. These measures aim to prevent future occurrences of similar tragedies and to restore public trust in the safety and efficacy of Indian-made pharmaceuticals. The government has also pledged to provide compensation and support to the victims and their families impacted by the Coldrif cough syrup incident.
The arrest of the pharma company owner underscores the potential consequences of neglecting quality control and regulatory compliance within the pharmaceutical industry. It sends a strong message that the safety of consumers must be prioritized above all else, and that those who compromise public health will be held accountable. The ongoing investigation and subsequent legal proceedings are expected to serve as a deterrent to other companies that may be tempted to cut corners or disregard safety standards.
The repercussions of the Coldrif cough syrup tragedy are likely to extend beyond the immediate legal and regulatory actions. The incident has sparked a broader debate about the need for greater transparency, accountability, and ethical conduct within the Indian pharmaceutical industry. It has also highlighted the importance of robust pharmacovigilance systems to detect and respond to adverse drug reactions promptly.
Image Source: Google | Image Credit: Respective Owner