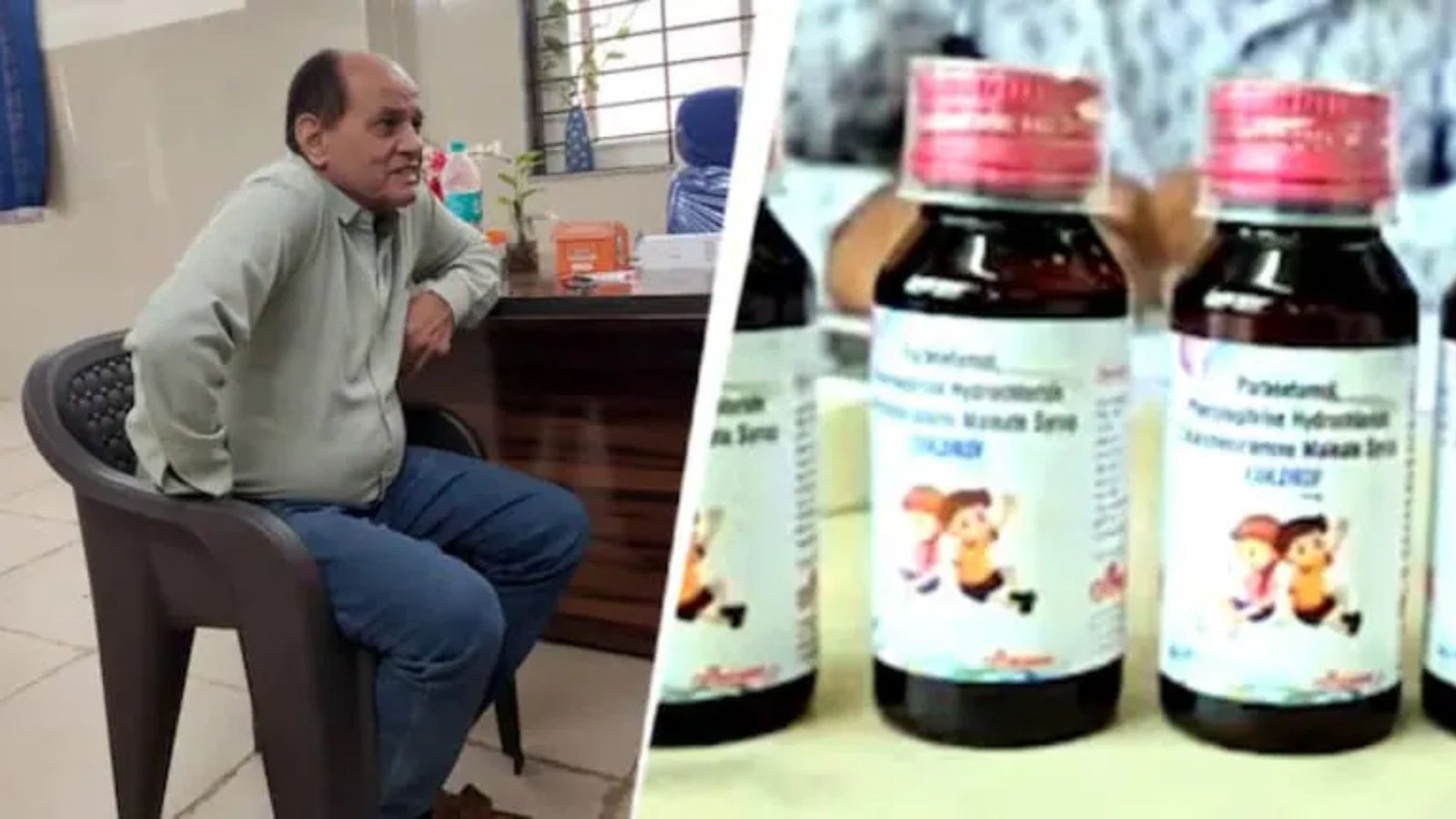
In a significant development in the investigation into cough syrup-related deaths, police have informed a court that a doctor allegedly received a 10% commission from the syrup’s manufacturer. The case revolves around the tragic deaths allegedly linked to contaminated cough syrup, raising serious concerns about pharmaceutical regulation and ethical practices within the medical profession.
Details of the Allegations
According to police reports presented in court, the doctor is suspected of prescribing the cough syrup in exchange for financial incentives from the manufacturing company. This arrangement, if proven, would constitute a grave breach of medical ethics and potentially implicate the doctor in the chain of events leading to the fatalities. Authorities are meticulously gathering evidence to substantiate these claims and determine the extent of the doctor’s involvement.
The ongoing investigation aims to uncover the full scope of the alleged commission scheme and its impact on the prescription and distribution of the implicated cough syrup. Investigators are examining financial records, communication logs, and witness testimonies to build a comprehensive case.
Wider Implications
This case highlights the urgent need for stringent oversight of pharmaceutical manufacturing processes and distribution channels. The allegations of corruption and compromised medical judgment underscore the potential dangers when financial incentives overshadow patient safety. Public health officials are now under increased pressure to strengthen regulatory frameworks and ensure greater accountability within the pharmaceutical industry and the medical community.
The revelations have sparked public outrage, with calls for swift and decisive action against those found responsible. Families of the victims are demanding justice and assurances that such tragedies will not be repeated. Consumer advocacy groups are advocating for stricter quality control measures and enhanced transparency in the pharmaceutical sector.
Legal Proceedings and Next Steps
The court is currently reviewing the evidence presented by the police and considering further legal action. The doctor in question may face charges related to negligence, criminal conspiracy, and violation of medical ethics. The manufacturer could also face severe penalties if found guilty of producing and distributing substandard or contaminated products.
As the legal proceedings unfold, the focus remains on uncovering the truth and holding those responsible accountable for their actions. This case serves as a stark reminder of the importance of ethical conduct, regulatory compliance, and patient safety in the pharmaceutical industry and the medical profession. The outcome of this case could have far-reaching implications for the future of pharmaceutical regulation and medical ethics in India.
The investigation is ongoing, and further details are expected to emerge as the case progresses. The authorities have assured the public that they are committed to pursuing all leads and ensuring that justice is served.
Image Source: Google | Image Credit: Respective Owner