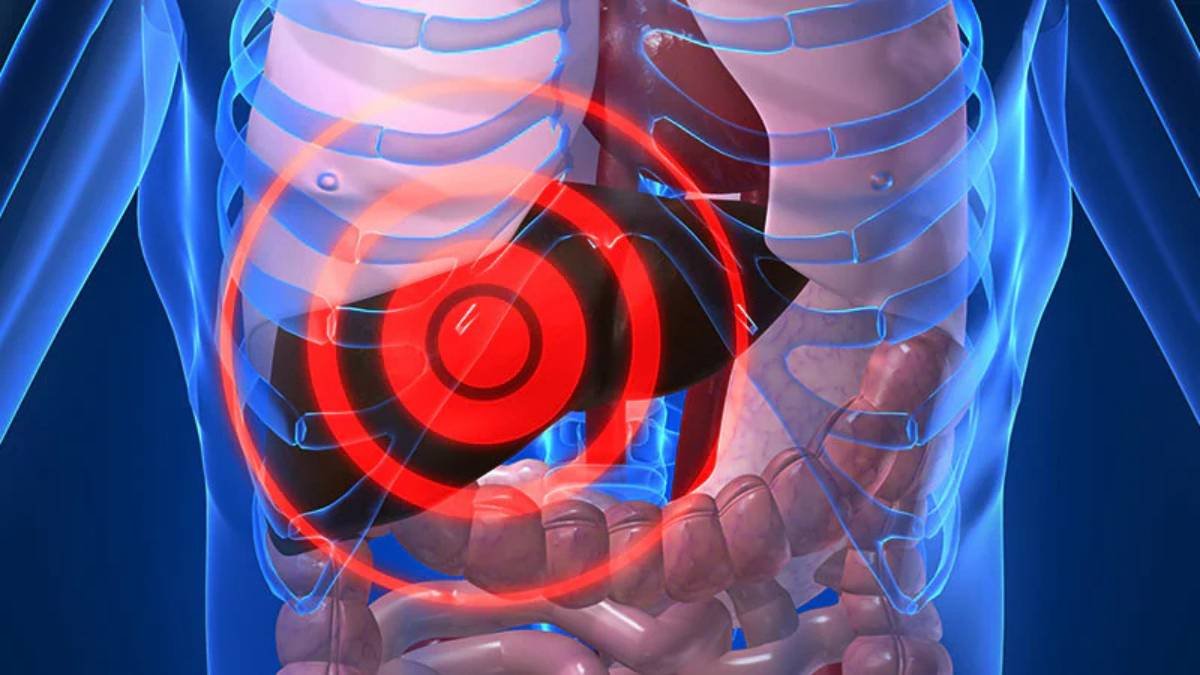
Noninvasive tests could potentially track a patient’s response to semaglutide treatment for metabolic dysfunction-associated steatohepatitis (MASH), according to a recent report by Medscape. MASH, formerly known as nonalcoholic steatohepatitis (NASH), is a progressive form of nonalcoholic fatty liver disease (NAFLD) characterized by liver inflammation and damage, often leading to cirrhosis, liver failure, and liver cancer. The increasing prevalence of obesity and type 2 diabetes has fueled the rise in MASH cases worldwide, creating a significant unmet medical need for effective therapies.
Currently, liver biopsy remains the gold standard for diagnosing and monitoring MASH. However, it is an invasive procedure associated with risks, including pain, bleeding, and sampling errors. Therefore, the development of reliable noninvasive biomarkers is crucial for improving patient management and accelerating drug development in this field. Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, has shown promise in clinical trials for reducing liver fat and improving liver enzymes in patients with MASH.
Potential Biomarkers for Semaglutide Response
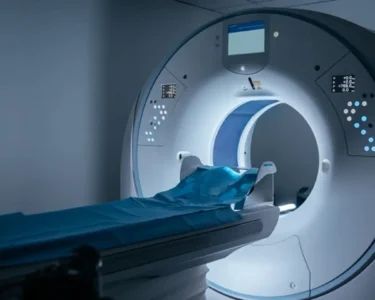
The Medscape report highlights several noninvasive tests that are being investigated as potential biomarkers for tracking semaglutide response. These include serum biomarkers, such as enhanced liver fibrosis (ELF) score, FibroTest, and NAFLD Fibrosis Score (NFS), as well as imaging techniques like magnetic resonance imaging-proton density fat fraction (MRI-PDFF) and vibration-controlled transient elastography (VCTE), also known as FibroScan. These tests aim to assess liver fat content, inflammation, and fibrosis, which are key features of MASH.
MRI-PDFF is considered a highly accurate method for quantifying liver fat, while VCTE measures liver stiffness, an indicator of fibrosis. Changes in these parameters during semaglutide treatment may correlate with histological improvements observed in liver biopsies. Furthermore, artificial intelligence (AI) and machine learning algorithms are being applied to integrate data from multiple noninvasive tests and predict treatment response with greater accuracy. These advanced tools have the potential to personalize treatment strategies and identify patients most likely to benefit from semaglutide.
The ongoing research in this area is focused on validating these noninvasive biomarkers in larger cohorts of patients with MASH and correlating them with long-term clinical outcomes. If successful, these tests could replace or reduce the need for liver biopsies, making it easier and safer to monitor treatment response and manage patients with MASH. The development of reliable noninvasive biomarkers represents a significant step forward in the fight against this growing global health challenge, potentially improving the lives of millions affected by MASH.
Further research is also exploring the potential of combining different biomarkers to create a more comprehensive assessment of liver health. This multi-parametric approach could provide a more accurate and nuanced understanding of the disease process and treatment response, leading to more effective and personalized therapies for MASH.
Image Source: Google | Image Credit: Respective Owner